 
Fermions include elementary particles such as quarks (the constituent particles of protons and neutrons), electrons and neutrinos.The Pauli exclusion principle governs the behavior of all fermions ( particles with half-integer spin), while bosons ( particles with integer spin) are not subject to it.More technically, it states that the total wave function for two identical fermions is antisymmetric with respect to exchange of the particles.In addition, the discovery of subatomic particles has shown that atoms can be divided into smaller parts.Dalton hypothesized this was due to the differences in the mass and complexity of the gases' respective particles.Knowing that a gas is composed of small atomic and molecular particles, it is natural to try to explain properties of the gas from a microscopic point of view.Although physicists discovered that the so-called "indivisible atom" was actually a conglomerate of various subatomic particles, the concept of atoms is still important because they are building blocks of matter and form the basis of chemistry.For example, when particles are destroyed, equal numbers of positive and negative charges are destroyed, keeping the net amount of charge unchanged. 
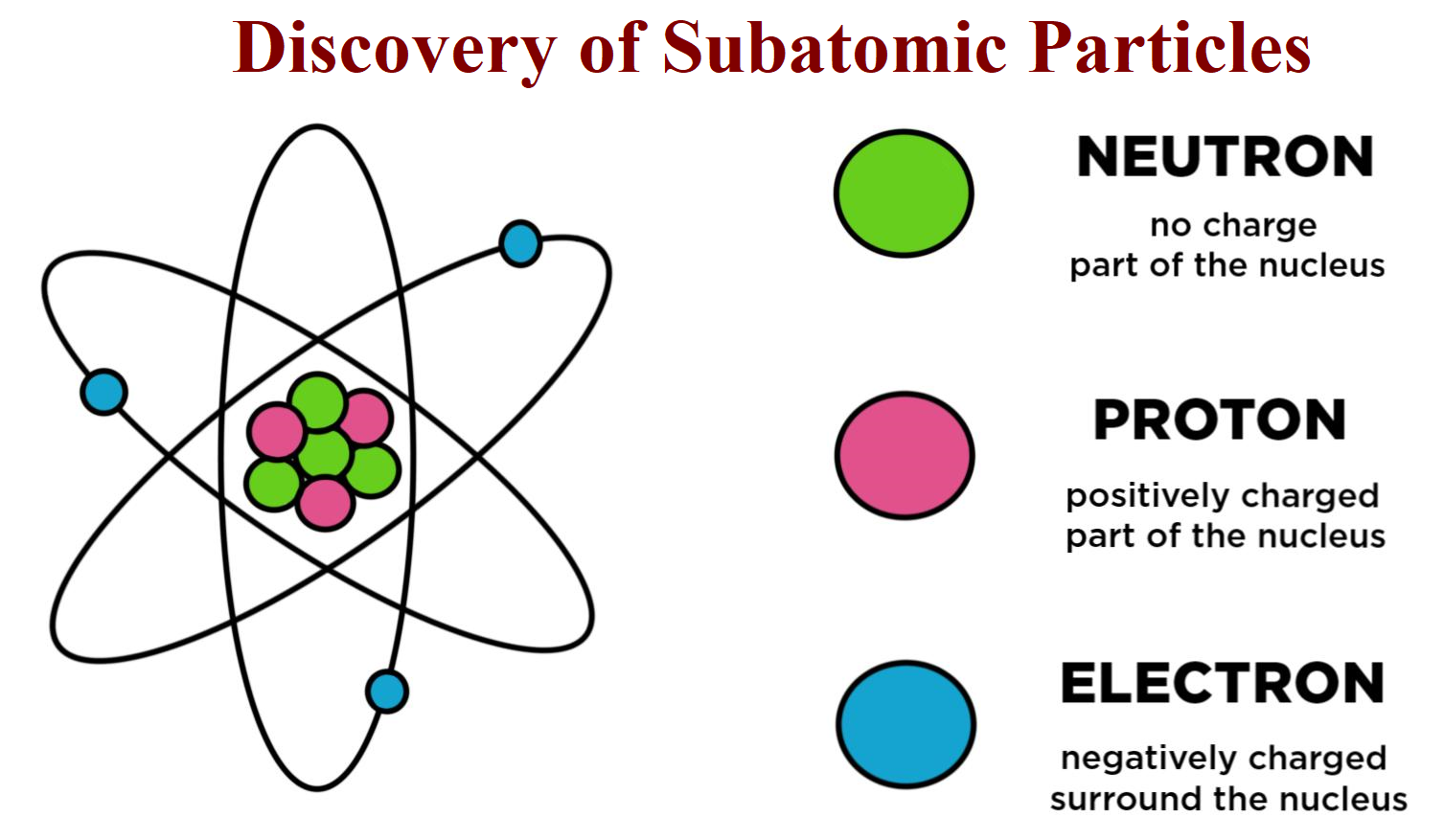
Electric charge is carried by subatomic particles such as electrons and protons, which can be created and destroyed.In both instances, charged particles will experience a force when in the presence of other charged matter.Static Electricity, Charge, and the Conservation of Charge.The behavior of the subatomic particles (electrons, protons, neutrons, photons, and others) that make up all forms of matter can often be satisfactorily described only using quantum mechanics.These particles had a similar mass as protons, but did not have the same characteristics as protons.In 1932, James Chadwick showed uncharged particles in the radiation he used.His explanation for this was the existence of a neutrally charged particle within the atomic nucleus.Though originally viewed as a particle that cannot be cut into smaller particles, modern scientific usage denotes the atom as composed of various subatomic particles.Modern scientific usage denotes the atom as composed of constituent particles: the electron, the proton and the neutron.Like the early philosophers, Dalton's theories were not popularly accepted for much of the 19th century, but his ideas have since been accepted, with amendments addressing subatomic particles and the interconversion of energy and mass.Democritus proposed that different types and combinations of these particles were responsible for the various forms of matter.The Greeks called these particles atomos, meaning indivisible, and the modern word "atom" is derived from this term. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
